Sensitive cavity-enhanced absorption spectrometry for gas-phase chemical kinetics
Technical Details
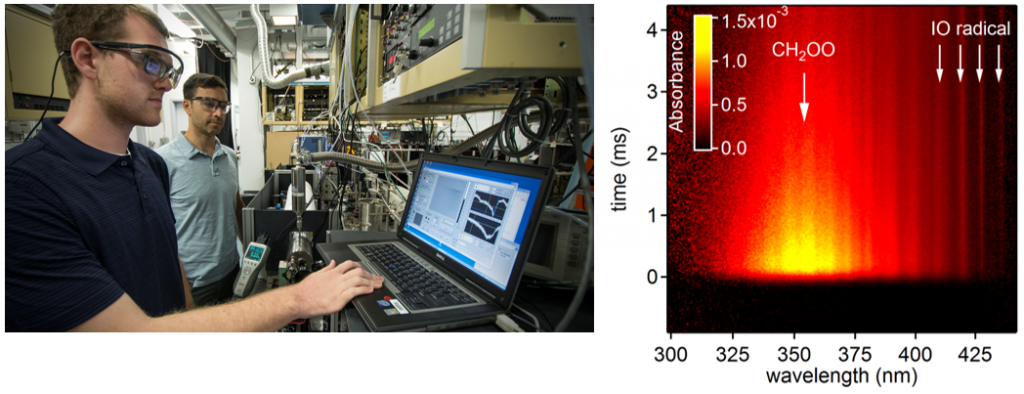
Absorption of photons from the IR to the UV spectral range can selectively probe ro-vibrational and electronic transitions of molecules, allowing for characterization of chemical reaction products and intermediates. Direct absorption spectroscopy is a ubiquitous experimental tool for the study of condensed phases, i.e., liquids, rare gas matrices, and solids. However, in gases the concentrations of reactive molecules are too low to absorb enough light for sensitive detection. Consequently, multi-pass methods are used to provide long effective sample path lengths and increase sensitivity. At the CRF, we have developed a new time-resolved broad-band cavity-enhanced absorption spectrometer for gas-phase chemical kinetics studies.
Our spectrometer is incorporated into a laser photolysis reactor in which a laser pulse initiates reactions at repetition rates 1 – 10 Hz. Continuous probe light from a simple Xe arc lamp builds up between two reflective end mirrors, resulting in path length enhancement of 50x – 100x over the entire near-UV to Visible spectral range. The optical buildup cavity output is analyzed by a custom spectrometer, which maps the wavelength and time dimensions spatially onto a CCD camera. Typically, long-pass absorption experiments are done by scanning either the probe wavelength or the time delay, both of which are slow and labor-intensive. By contrast, our TR-BB-CEAS apparatus records the full spectral time evolution simultaneously for each photolysis laser shot, resulting in rapid multiplexed kinetics measurements with microsecond time resolution.
Key Contributions
- Measured the UV absorption spectra of several prototypical Criegee intermediates: CH2OO, syn– and anti-CH3CHOO, and CH3C(OO)CHCH2
- Used the strong UV absorption bands of Criegee intermediates to probe directly the kinetics of their gas-phase reactions with water, H2O dimer, and key trace atmospheric compounds
Partners
- Marsha I. Lester, University of Pennsylvania
- Carl Percival, Jet Propulsion Laboratory
- Andrew Orr-Ewing, University of Bristol
- Dudley Shallcross, University of Bristol
- Rebecca Caravan, Argonne National Laboratory
PIs: Leonid Sheps, Craig A. Taatjes
