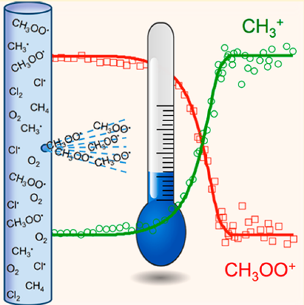
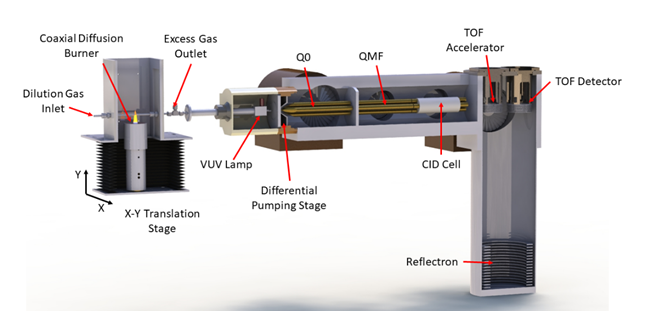
At the CRF, researchers apply the power of mass analysis to investigate the details of complex chemical reaction networks such as those that drive combustion, autoignition, and tropospheric autoxidation.” Development and application of synchrotron photoionization methods have proved particularly powerful, and the CRF has benefited from a close collaborative relationship with researchers at Lawrence Berkeley National Laboratory’s Advanced Light Source (https://als.lbl.gov/). Our scientists have developed new mass spectrometers and reactors that have pushed the frontiers of experimental reaction kinetics and combustion chemistry.