Although a powerful technique, analyzing molecules based on mass does not always enable us to identify specific molecular structures. Photoionization spectroscopy can yield isomer-specific information from mass spectrometry, but as the molecules grow larger in the chemistry leading to soot formation, for example, the photoionization spectra are no longer diagnostic. CRF researchers have used another mass spectrometric method, tandem mass spectrometry for such challenging applications. To implement tandem mass spectrometry, CRF researchers sample molecules from a reactive environment (e.g., a flame) directly into an atmospheric pressure photoionization (APPI) source where they are ionized using 10.0 eV photons from a continuous vacuum-ultraviolet (VUV) Kr discharge lamp. This photon energy is sufficient to ionize the targeted aromatic and highly oxygenated species important in particle formation. The resulting ions, when mass selected and accelerated into the argon-filled collision-induced dissociation (CID) cell, can fragment into a neutral and an ion fragment. Those fragment mass spectra indicate which components exist in the original molecule. For example, as illustrated below, C32H20 molecules observed in samples from a flame are shown to be a mix of C16H20 dimers and mixed (C14H10 + C18H10) clusters.
Technical Details
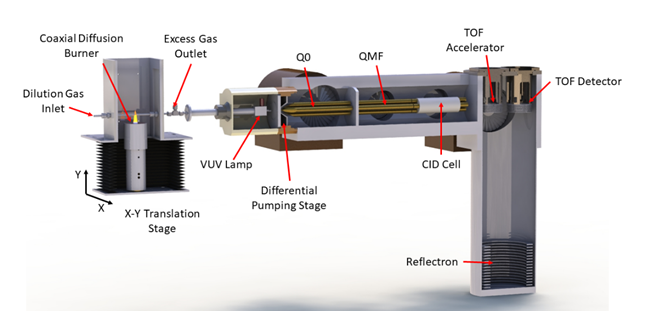
Figure 1 Diagram of a tandem mass spectrometric analyzer used to investigate the composition of a flame. Molecules are swept from the burner (far left) into the ionization region and ionized by a VUV lamp. The resulting ions can be mass-analyzed in the quadrupole mass spectrometer Q0 and selected masses can be passed through the quadrupole mass filter (QMF) and accelerated into a cell filled with argon gas. Collisions with the argon can break the initial ion into fragment ions, and a second mass spectrometer measures the mass of the fragments using time of flight (TOF) methods (far right). The efficiency of this fragmentation depends on the energy to which the initial ions were accelerated, and the fragmentation pattern reveals the constituents of the original ion (Source: https://doi.org/10.1021/acs.jpca.8b08947)
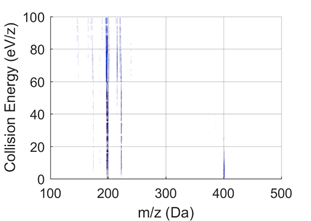
Figure 2 Graph: Tandem mass spectrometry showing fragmentation of initial ions of mass/charge ratio (m/z) = 404.157 (C32H20) sampled from a fuel-rich flame. Fragmentation of the ions into m/z = 202.078 (C16H10) at low collision energies is visible. The fragmentation reveals that C32H20 also contains (C14H10 + C18H10) dimers. Right graph: High-resolution integrated mass spectra at low (from 0-10 eV) collision energies (Source: https://doi.org/10.1515/zpch-2020-1638)
Key Contributions
- PAH dimers are not important in soot nucleation
- Aliphatically substituted PAH observed in sooting flames
Partners: Musahid Ahmed, Lawrence Berkeley National Laboratory
PI: Nils Hansen