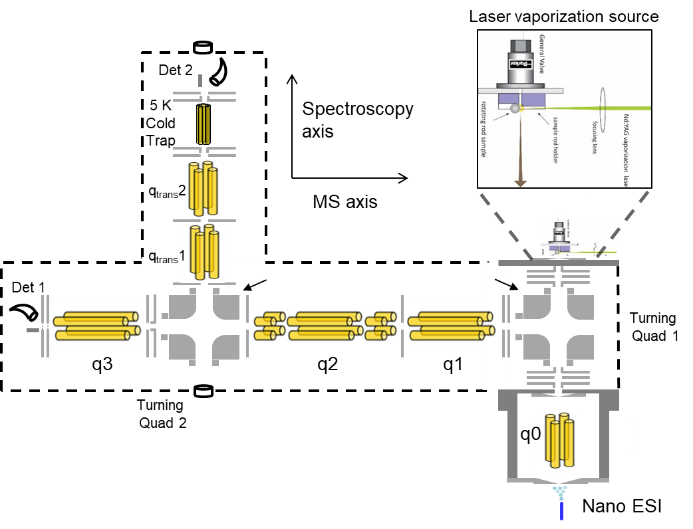
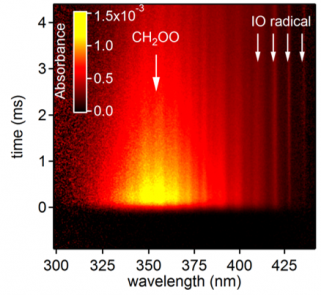
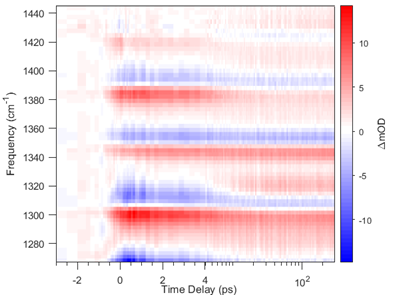
The Combustion Research Facility has a long history of leadership in laser diagnostics, developing and characterizing optical laser techniques to detect and quantify important gas-phase species, temperature, and particulate matter in temporally and spatially resolved measurements. Research in spectroscopic methods underlies this leadership. CRF researchers are developing capabilities that interrogate molecular species using radiation from the microwave to the X-ray region, probing environments that stretch from isolated molecular ions to species sampled from high-pressure high-temperature reactors.