Photoelectron Photoion Coincidence Spectroscopy (PEPICO): A New Tool for Chemical Physics of Complex-Forming Reactions
Technical Details
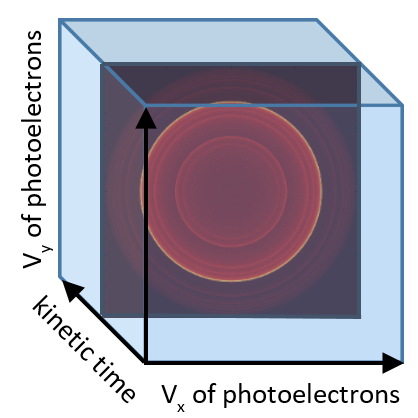
Nature is replete with complex-forming chemical reactions, such as OH + CH3-CH=CH2 (propene). The potential energy surface for these reactions is characterize by multiple deep wells (representing different isomers of the OH-propene adduct) with many available bimolecular product channels. To probe such reactions, we developed a new use for a classic technique in chemical physics: photoelectron photoion coincidence spectroscopy (PEPICO). After initiating a chemical reaction with a laser pulse, we use single vacuum ultraviolet photons to ionize all species (reactants, intermediates, and products). We detect both the cations and the electrons formed from this ionization using ion and electron imaging (a technique created at the CRF by David W. Chandler and Paul Houston). The results provide photoelectron spectra that arise from each unique mass-to-charge ratio from the chemically reacting system under study. These photoelectron images (Fig. 1) provide unique fingerprints to identify which chemical isomers are produced at each molecular mass and allow us to follow reactions as a function of kinetic time.
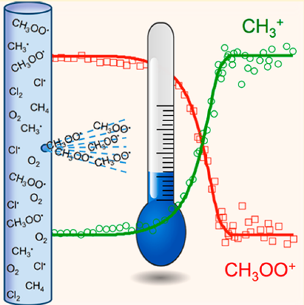
Our approach provides many new tools to the chemical physicist, such as a Radical Thermometer that measures the internal energy of reactive free radicals, as depicted in Figure 2. Our new instrument has already revealed an unexpected product channel in the photodissociation of SO2 that may impact our understanding of Earth’s early atmosphere.
Key Contributions
- Multiplexed probing of time-resolved chemical reactions with isomeric selectivity
- Breaking through the false coincidence barrier in coincidence spectroscopy
Partners
- Balint Sztaray, University of the Pacific
- Andras Bodi, Paul Scherrer Institute, Switzerland
- Patrick Hemberger, Paul Scherrer Institute, Switzerland
PI: David L. Osborn