The chemical reactivity of gases with solid surfaces is ubiquitous in natural and industrial energy transformation. Cooperative effects that couple gas phase chemistry with surface chemistry are critical for foundational understanding, but they prove challenging to probe experimentally and theoretically. Heterogenous catalysis is an ideal field in which to expose and isolate the fundamental chemical physics of these cooperative effects. We are pursuing a program to characterize gas-surface coupling, through chemically specific, temporally and spatially resolved probes of both reacting surfaces and the near-surface gas phase. The program combines linear and non-linear optical spectroscopies, universal photoionization mass spectrometry, and surface-specific spectroscopies at ambient pressures. Breakthrough coherent nonlinear spectroscopic imaging methods enable mapping of surface-gas exchange, which, combined with in operando surface spectroscopy, will correlate reaction rates with local surface structure, composition, and oxidation state during catalytic chemical transformations. These correlated measurements in self-organized oscillatory reaction systems will directly quantify how different surface domains communicate via chemical transport and are linked by chemical feedback. We have observed reactive intermediates above surfaces, which both establishes critical mechanistic links between surface and gas-phase reactivity and provides detailed species maps to inform microkinetic models. The long-term goal of this project is to elucidate the fundamental mechanisms of cooperative gas-surface chemistry, influencing DOE mission research in catalysis, synthesis, and energy transformation.
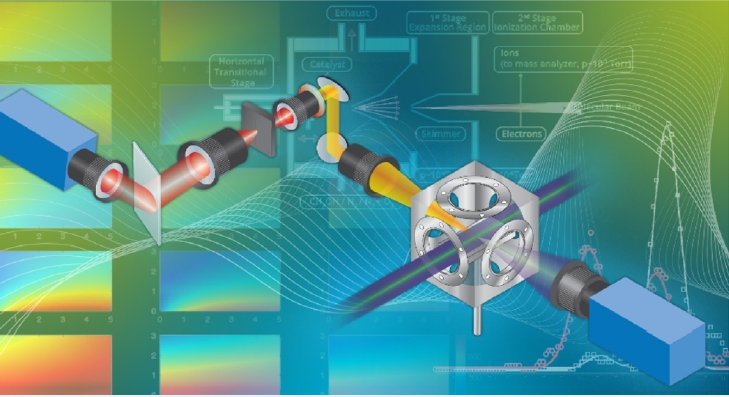
Figure 5 We image the gas phase immediately above a reacting surface using three complementary techniques: Planar laser induced fluorescence (lower right), Raman spectroscopy (upper left), and near-surface molecular beam mass spectrometry (center, background). Source: Sandia National Laboratories.
Partner
Prof. Johan Zetterberg, Lund University
PIs: Chris J. Kliewer, Nils Hansen, Jonathan H. Frank, Farid El Gabaly, and David L. Osborn