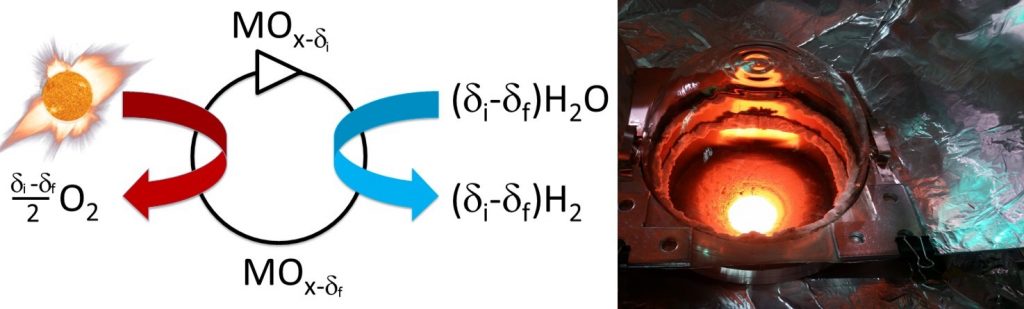
Figure 1 Shown here is the virtually accessible laser-heated stagnation flow reactor (LSFR) for characterizing redox chemistry of materials under extreme conditions. The LSFR is equipped with a solid-state laser to replicate the high energy flux and rapid heating rates that materials encounter in a concentrating solar reactor. The LSFR precisely measures both the amount of oxygen released from the material and the amount of hydrogen produced from splitting water during each step in the cycle. This unique facility is automated and can be operated remotely via a networked desktop computer from anywhere in the world.
Project
The Department of Energy supports the development of technology that produces hydrogen by directly splitting water using highly concentrated sunlight as process heat to power thermochemical cycles. Sandia’s LSFR facility was established to characterize redox active functionality in metal oxides exposed to extreme conditions, such as high radiant heat flux and high temperature. These conditions are commonly encountered in solar powered thermochemical cycles.
Figure 2 (Left) Schematic illustrating a generic two-step metal oxide reaction cycle. MOx is exposed to heat from concentrated solar energy causing spontaneous evolution of oxygen (left). The reduced oxide is then moved off sun and exposed to water vapor where oxygen stripped from the water molecule is reabsorbed by MOx liberating hydrogen. (Right) Looking into Sandia’s radiant cavity solar receiver reactor under operation. The temperature inside the receiver is 1450 °C.
Impact
Metal oxides that can efficiently and cost effectively conduct thermochemical H2 production at industrial scale remain elusive because optimizing the hydrogen production capacity and yield in non-stoichiometric oxides has thus far proved challenging. Redox active materials must not only release oxygen at temperatures accessible to concentrating solar power reactors, but split water from their oxygen-deficient state while maintaining both high capacity and high yield. Many factors that determine a material’s behavior are deeply rooted in its electronic structure. Understanding the fundamental underpinnings that link structure to desirable thermodynamic behavior is critical to developing new materials.
Technical Details
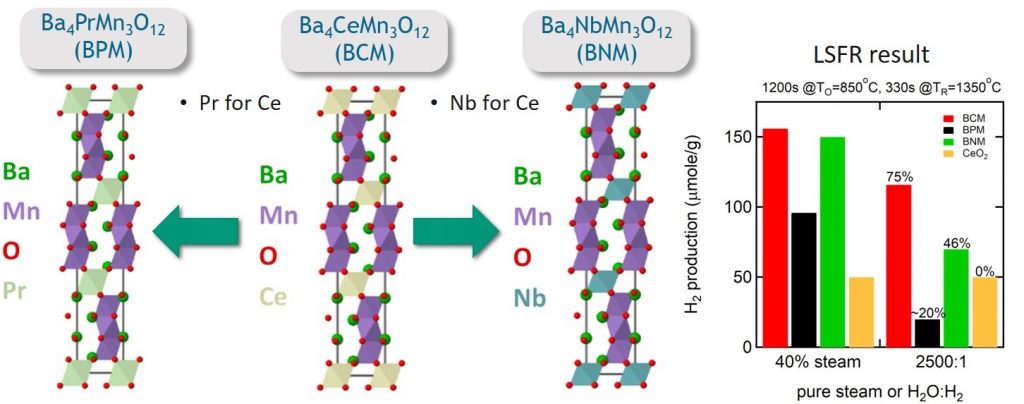
Layered perovskites are not commonly used in solar thermochemical water splitting cycles. However, we have discovered a family of complex perovskites that are B-site, Mn-based compounds (Ba4AMn3O12, A=Nd, Ce, Pr) with a cerium variant that outperforms all known perovskites tested for solar thermochemical water splitting functionality. We are interested in the behavior of Mn in the Ba4AMn3O12 family, and more broadly, in like-type Mn-based complex oxides. In these systems, the extent of electron sharing between Mn and the O ligand is highly malleable and greatly influences the material’s thermodynamic behavior. This can be seen by the stark differences between the capacity and hydrogen production yield in the Ba4AMn3O12 family upon substitution.
In addition, the polytypes accessible to this family of compounds are distinguished by the complexity of the Mn-O oligomer rearrangements within the layered structures. Fundamentally, we strive to understand to what extent electronic structure and crystallographic arrangements influence the enhanced water splitting thermodynamics and kinetics we observe in these complex perovskites. We believe that a comprehensive understanding of local structure in such compounds will lead to novel pathways for modifying and improving redox activity in perovskites.
Figure 3 Crystal structures for the Pr, Ce, and Nb substitutional variants found in the Ba4AMn3O12 family of compounds (A = Nb, Ce, or Pr). The Mn-O bonding environment consists of face-sharing octahedron trimers separated by rows of the other B-site cation. In the case of Pr and Ce that are both 4+ cations, Mn4+ is the oxidation state observed in the fully stoichiometric compound. In the niobium variant, Nb5+ donates an additional electron into the trimer and lowers the formal oxidation state of Mn in the system. The impact of these elemental substitutions can be seen in the bar graph where overall hydrogen production capacities measured in the LSFR at 40% steam changes with Ce > Nb > Pr. The production capacities in mixtures of steam and hydrogen (aka, high yield) are similarly affected. Solar thermochemical water splitting technology is commercially viable only when the redox active materials maintain production capacity at high yield.
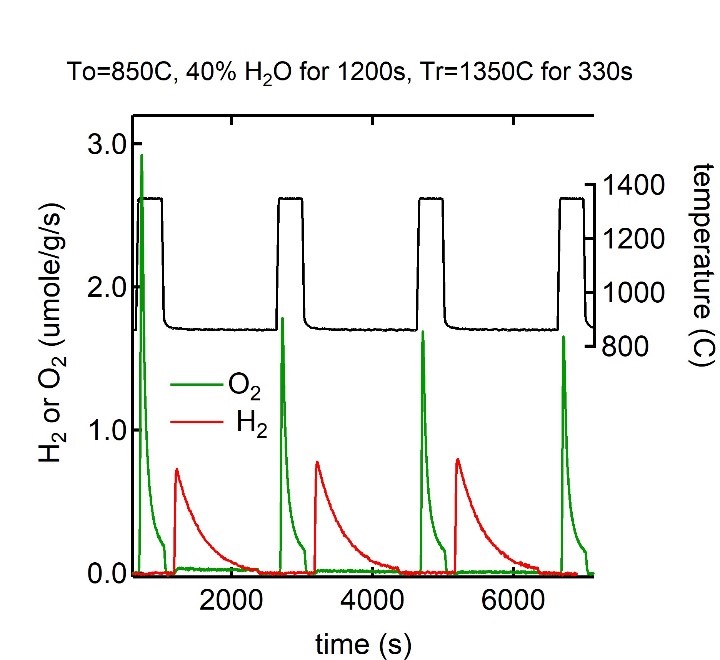
Figure 4 H2 and O2 production rates measured in the LSFR during three successive redox cycles on a particular perovskite material. Experimental conditions are listed above graph. The material is subject to heating rates greater than 15 °C per second, and overall temperature swings of 500 °C, in each cycle. The amount of H2 and O2 produced per a cycle is used to establish the water splitting efficacy of non-stoichiometric, non-volatile metal oxides used in two-step thermochemical cycles.
PI: Anthony McDaniel