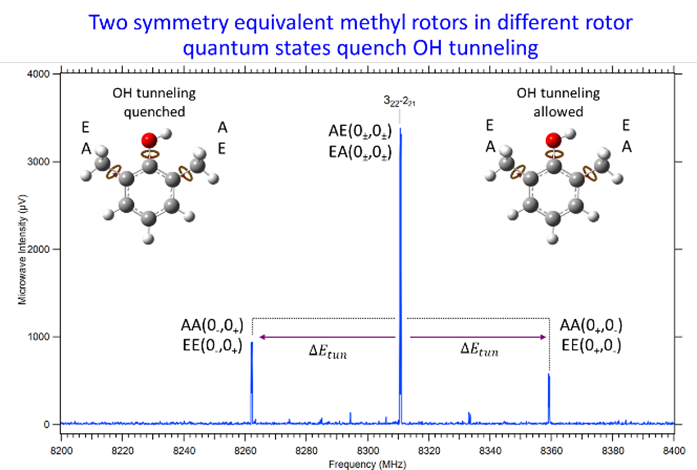
Tim Zwier, Judit Zádor and Blair Welsh from the CRF, with colleagues Sven Herbers from the Université Paris Est and Edwin L Sibert III from University of Wisconsin Madison, published a paper on “Methyl Rotor State-Dependent Quenching of OH Tunneling in 2,6-Dimethylphenol” in “The Journal of Physical Chemistry Letters”.
Tunneling is a purely quantum process where a particle moves through a barrier due to its wave nature. This phenomenon is not just an interesting curiosity observed in small molecules but is also used by some enzymes to accelerate reactions, especially hydrogen transfers, by enabling quantum tunneling rather than relying solely on classical thermal activation. Large kinetic isotope effects—differences in reaction rates between hydrogen and deuterium—often signal tunneling, as lighter isotopes tunnel more easily. Tunneling rates are highly sensitive to the details of the potential barrier, which depend on the local molecular environment. In this work the researchers used a combination of chirped-pulse Fourier transform microwave spectroscopy and advanced theory to investigate tunneling in 2,6-dimethylphenol, a molecule with two methyl groups adjacent to an OH group that undergoes tunneling. The complex tunneling regime is studied in enough detail to quantify the influence of the methyl states adjacent to the tunneling site. The impact is significant: depending on the whether the two methyl rotors are in the same methyl torsional state or not, tunneling can be effectively turned “on” or “off.” This work provides detailed insight into how coupled internal motions within a molecule modulates quantum tunneling.
For more information: https://doi.org/10.1021/acs.jpclett.6c00128
